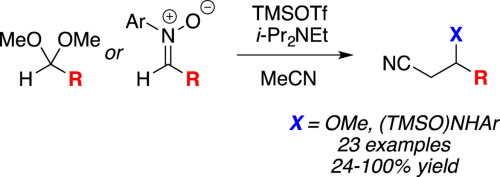
One-pot silyl ketene imine formation-nucleophilic addition reactions of acetonitrile with acetals and nitrones,Tetrahedron Letters - X-MOL
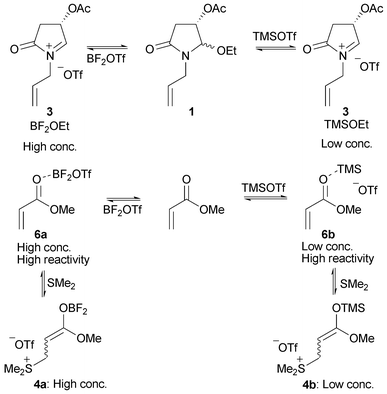
BF3·OEt2 and TMSOTf: A synergistic combination of Lewis acids - Chemical Communications (RSC Publishing)

Synthesis of Cyclic N‐Tosyliminocarbonates by Lewis Acid Catalyzed Allylic Substitution of Trichloroacetimidates - Grigorjeva - 2012 - European Journal of Organic Chemistry - Wiley Online Library
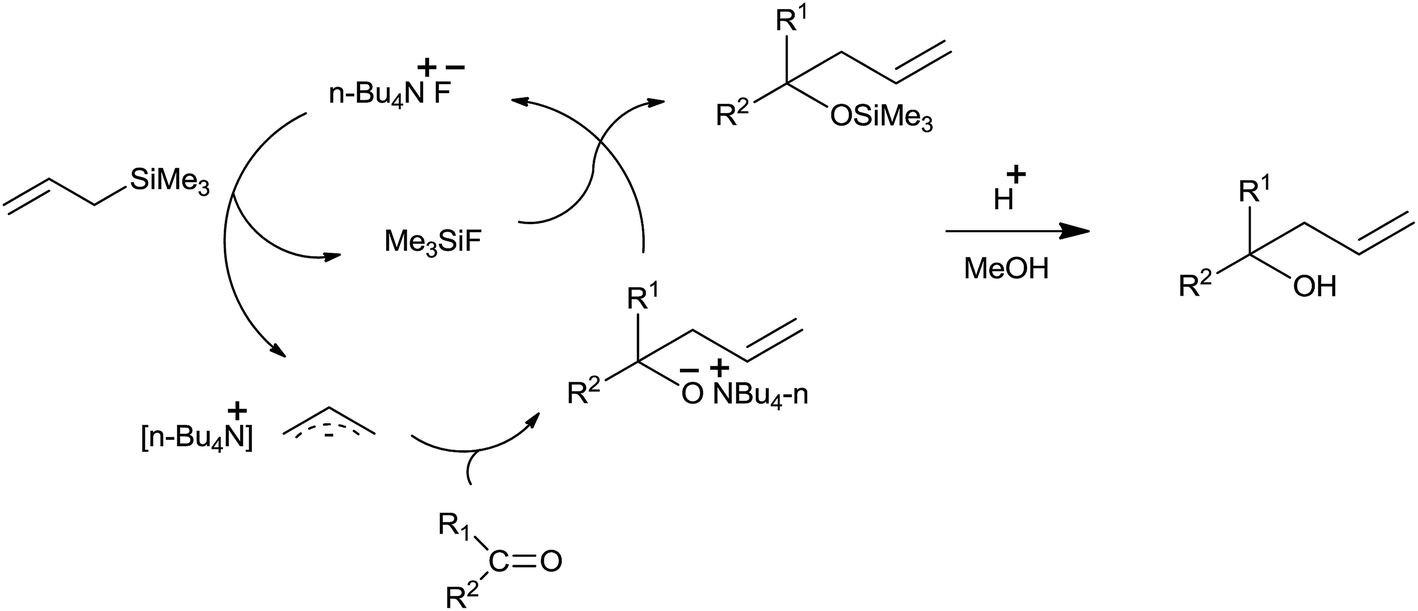
The remarkable journey of catalysts from stoichiometric to catalytic quantity for allyltrimethylsilane inspired allylation of acetals, ketals, aldehyd ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA27813B

Mukaiyama addition of (trimethylsilyl)acetonitrile to dimethyl acetals mediated by trimethylsilyl trifluoromethanesulfonate - ScienceDirect

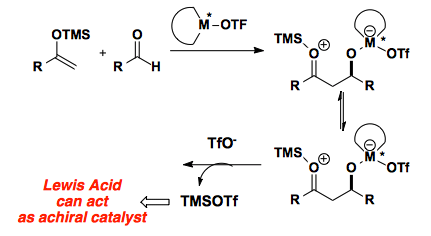
Report: New Reactivity Mediated by Trimethylsilyl Trifluoromethanesulfonate (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
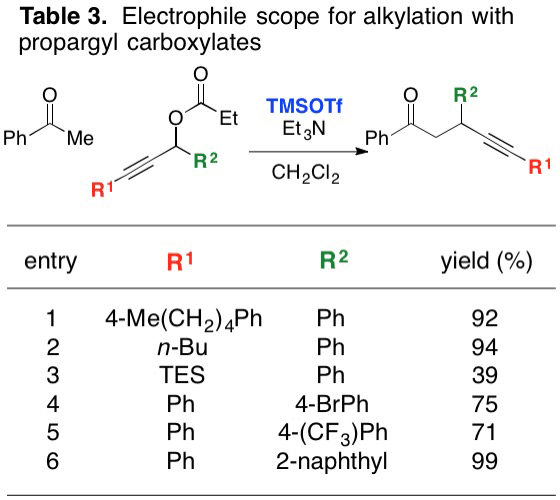
Report: New Reactivity Mediated by Trimethylsilyl Trifluoromethanesulfonate (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

Bi(OTf)3-, TfOH-, and TMSOTf-mediated, one-pot epoxide rearrangement, addition, and intramolecular silyl-modified Sakurai (ISMS) cascade toward dihydropyrans: comparison of catalysts and role of Bi(OTf)3. - Abstract - Europe PMC
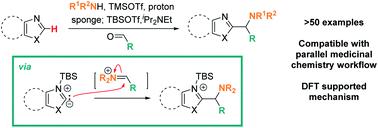
Lewis acid mediated, mild C–H aminoalkylation of azoles via three component coupling,Chemical Science - X-MOL

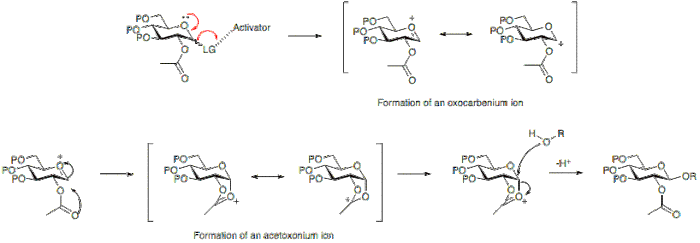
Glycosylation: The Direct Synthesis of 2‐Acetamido‐2‐Deoxy‐Sugar Glycosides - Beau - 2018 - European Journal of Organic Chemistry - Wiley Online Library
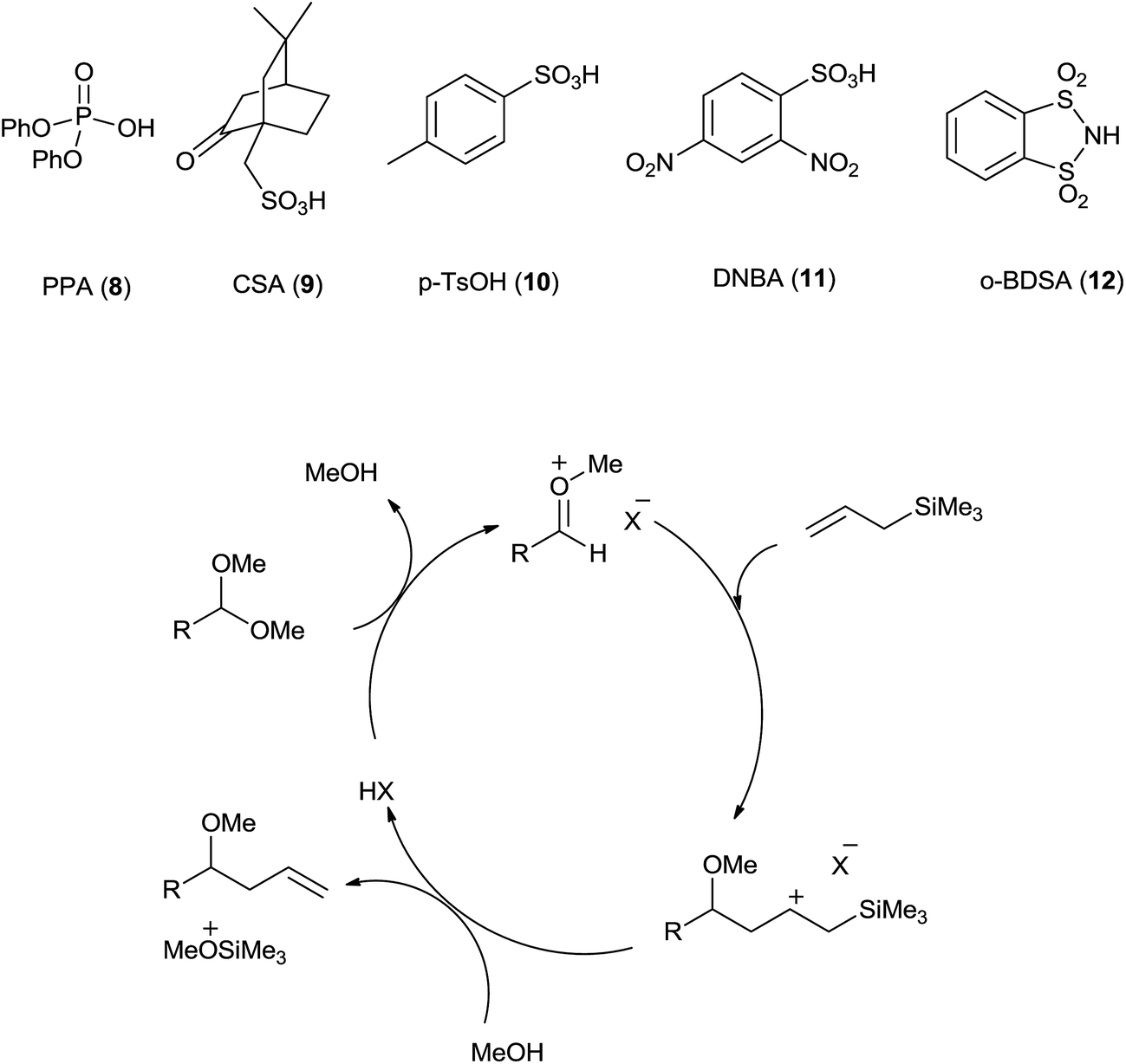
The remarkable journey of catalysts from stoichiometric to catalytic quantity for allyltrimethylsilane inspired allylation of acetals, ketals, aldehyd ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA27813B