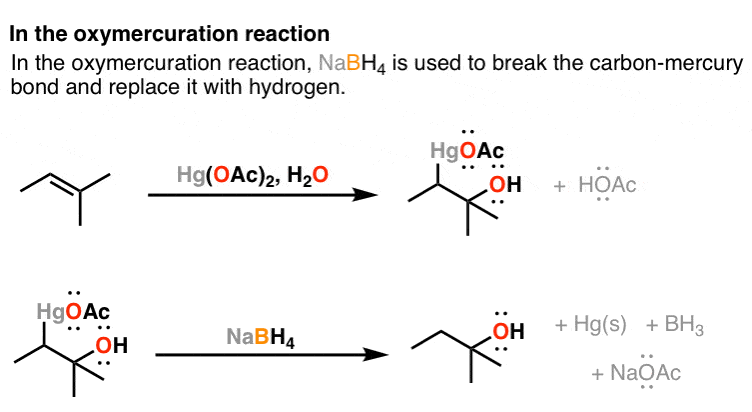
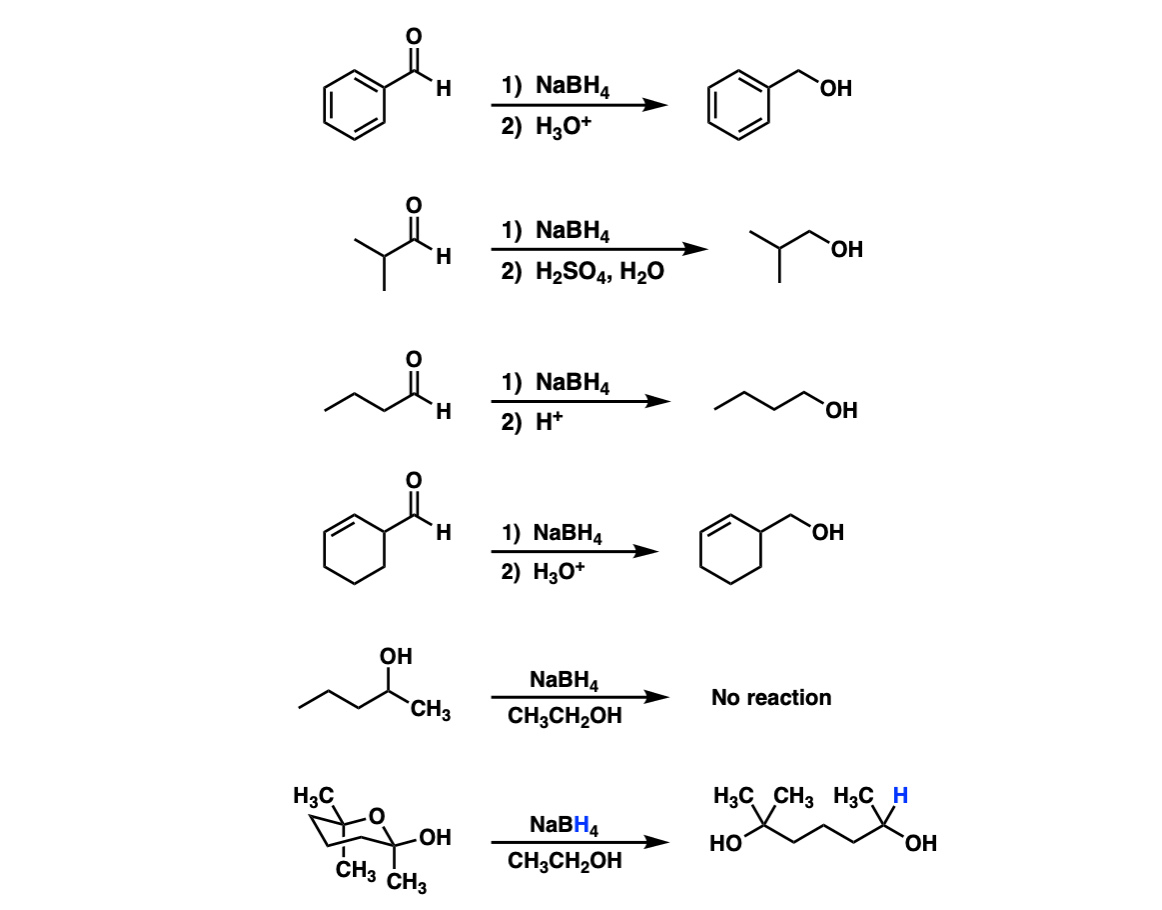
Enantioselective reduction of ketones with NaBH4/diglyme possibly catalysed by trialkyl borate: optically active sec-alcohols from prochiral ketones with catalytic (−)-menthol: autocatalysis option - ScienceDirect

NaBH4, LiAlH4, DIBAL Reduction Mechanism, Carboxylic Acid, Acid Chloride, Ester, & Ketones - YouTube

SOLVED:Introduction: The reduction of carbonyl compounds to alcohols using metal hydride reagents is common reaction_ Two of the most common metal hydride reducing agents are sodium borohydride (NaBH4) and lithium aluminum hydride (




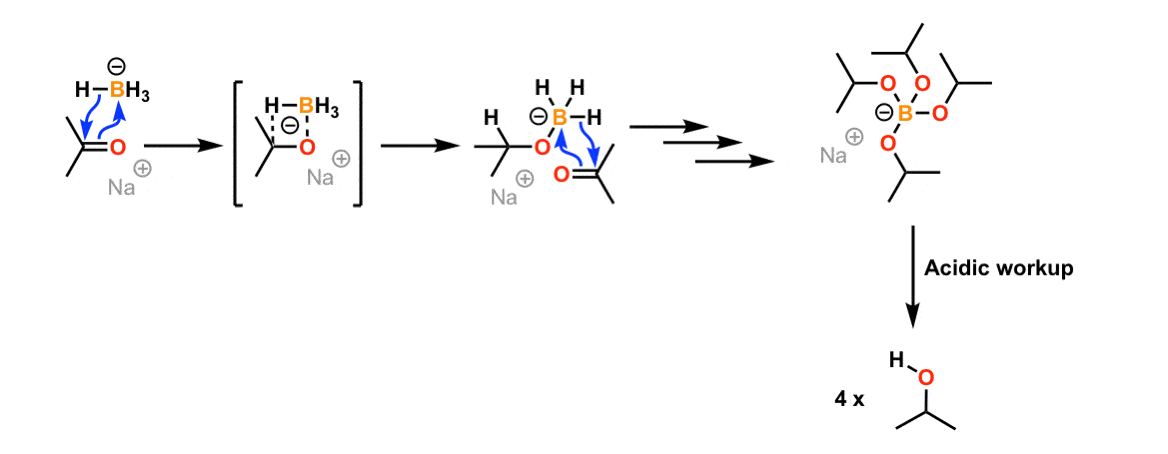



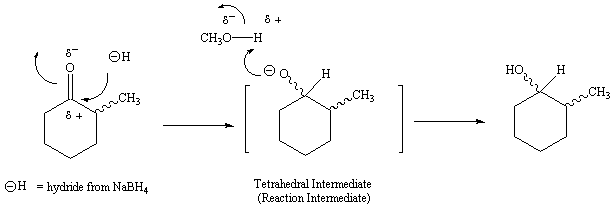

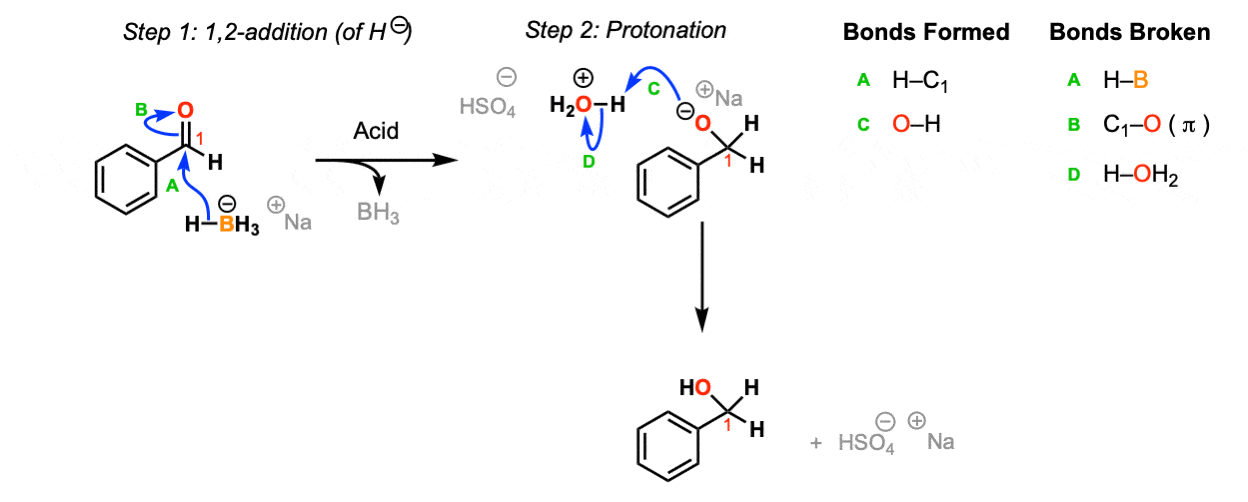
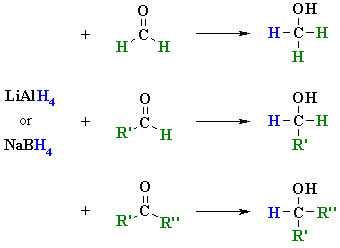


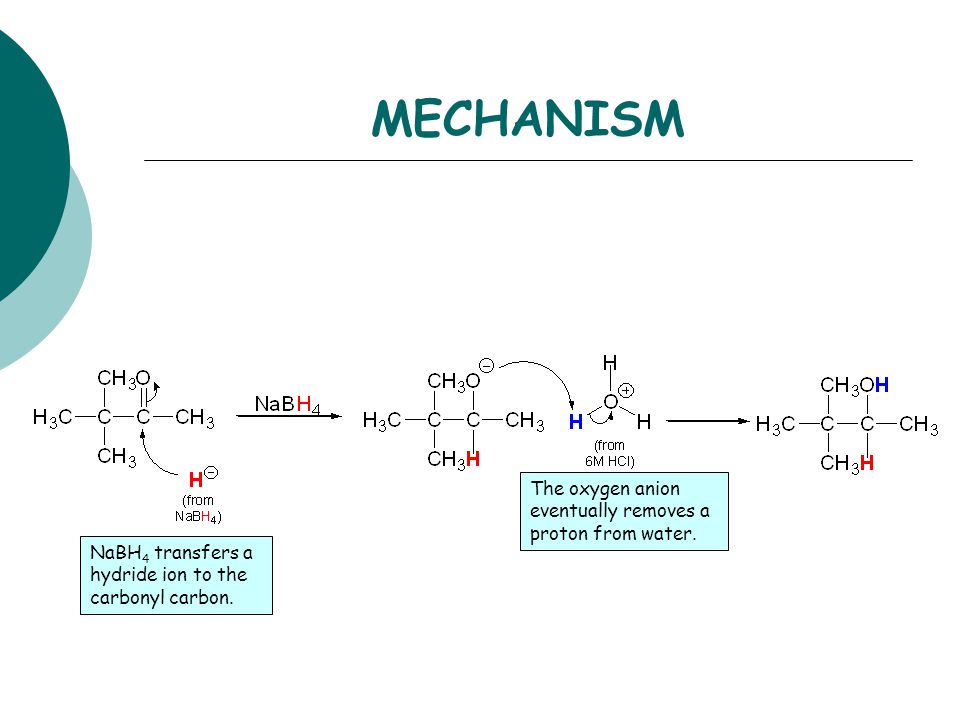
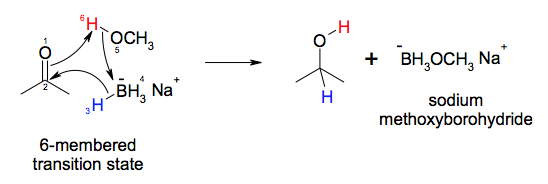



![Reduction of ketones [NaBH4] - ChemistryScore Reduction of ketones [NaBH4] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Reduction-of-ketones-NaBH42-1-1024x259.png)