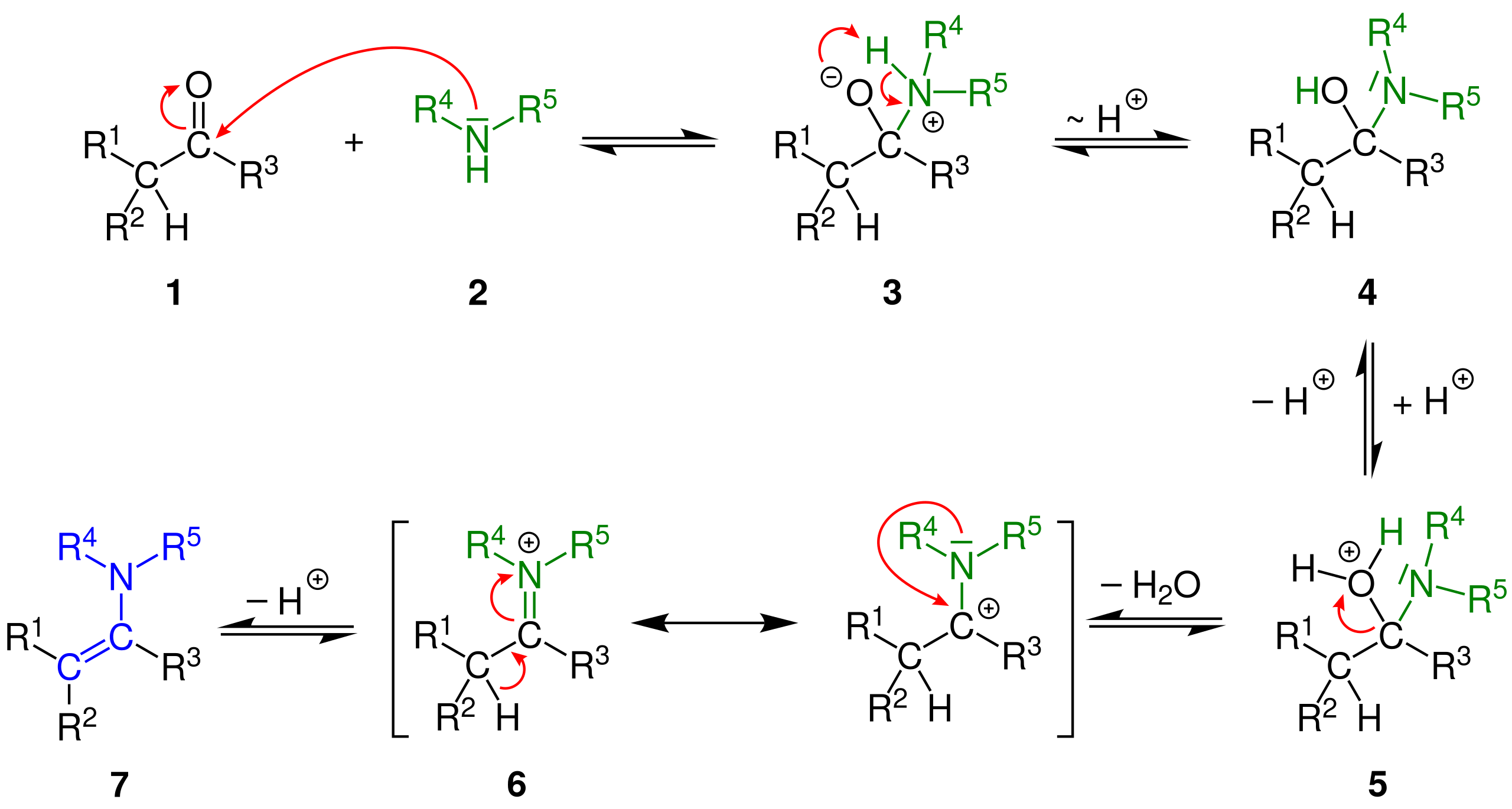
The Proline Enamine Formation Pathway Revisited in Dimethyl Sulfoxide: Rate Constants Determined via NMR. | Semantic Scholar

Reactive Enamines and Imines In Vivo: Lessons from the RidA Paradigm: Trends in Biochemical Sciences
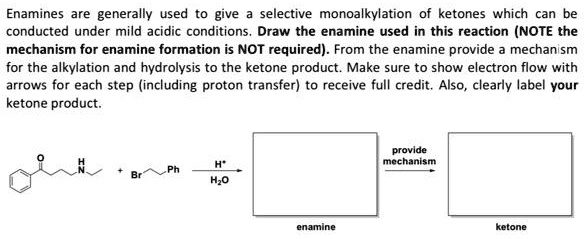
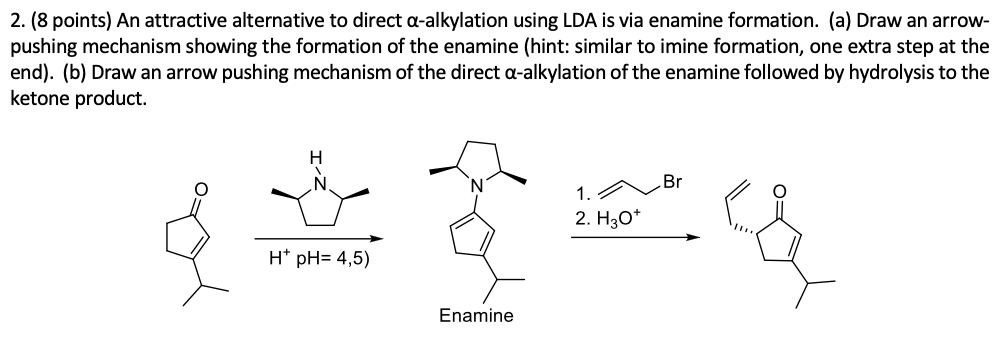
SOLVED:Enamines are generally used to give selective monoalkylation of ketones which can be conducted under mild acidic conditions_ Draw the enamine used in this reaction (NOTE the mechanism for enamine formation is

The Stork Enamine Synthesis used for Alkylation, Acylation, and Conjugate Addition | Organic chemistry, Organic chemistry study, Study chemistry
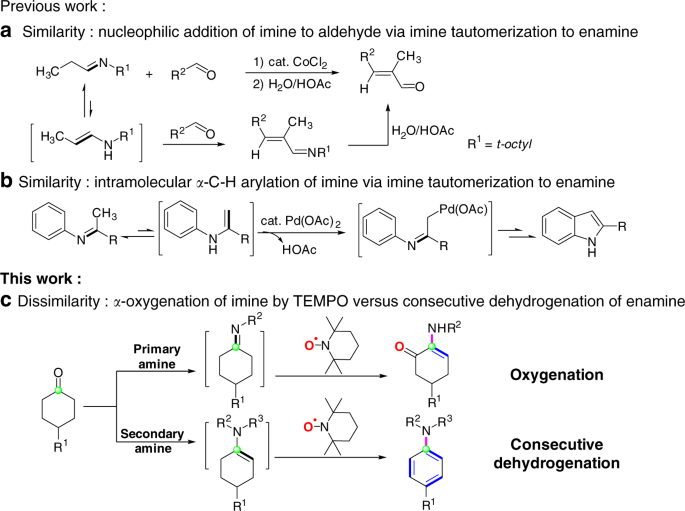
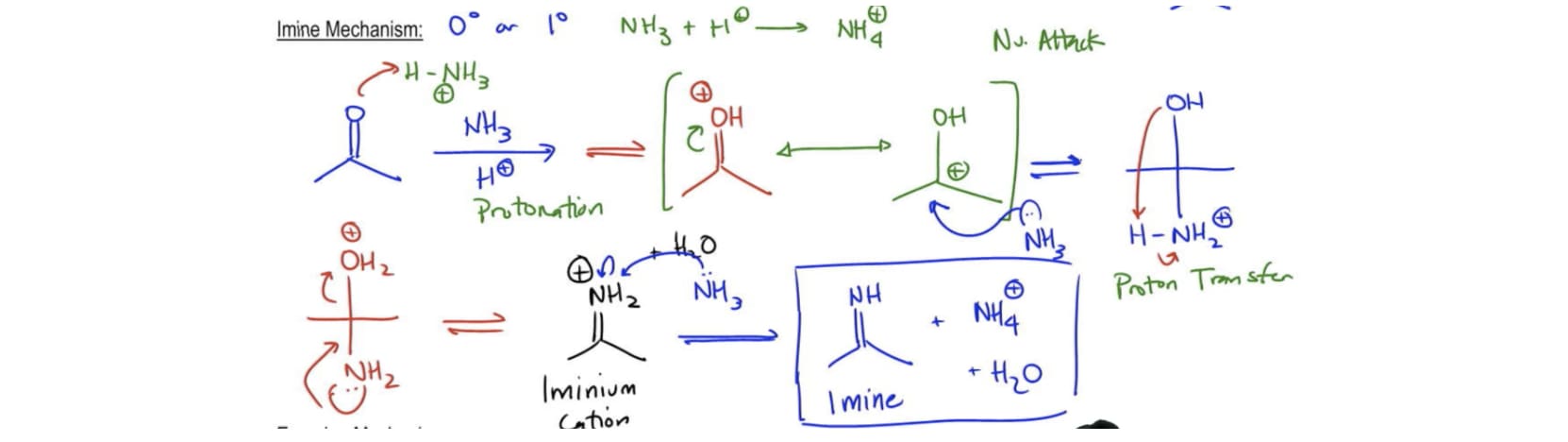
/chapter11/pages19and20/page19and20_files/enamine_examples.png)



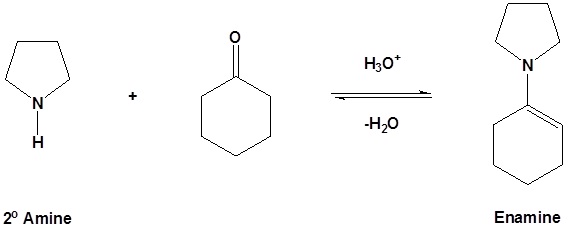

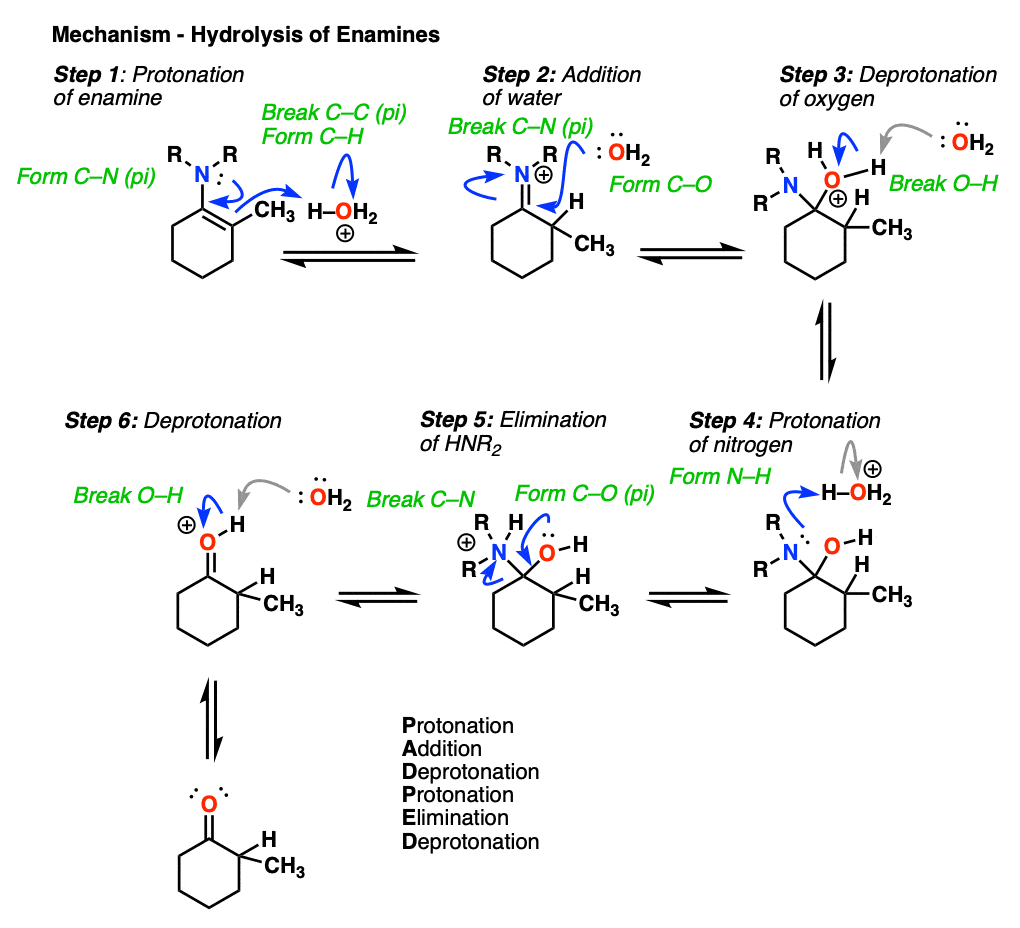


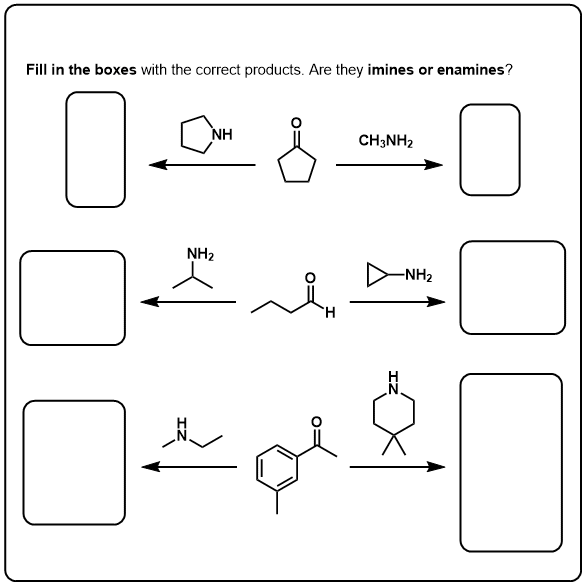
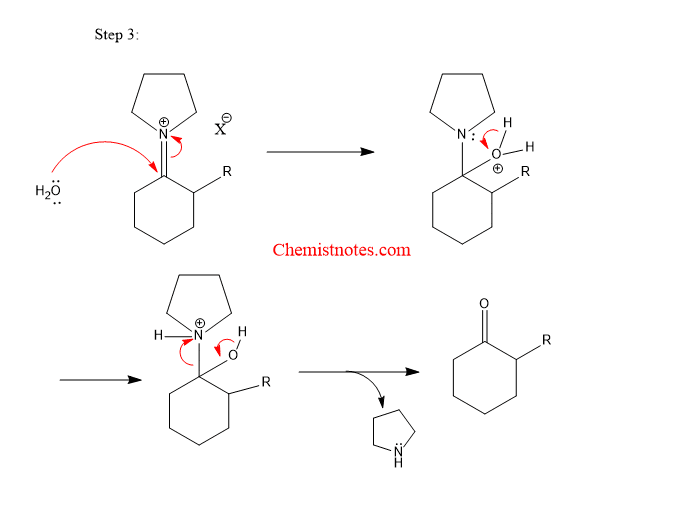


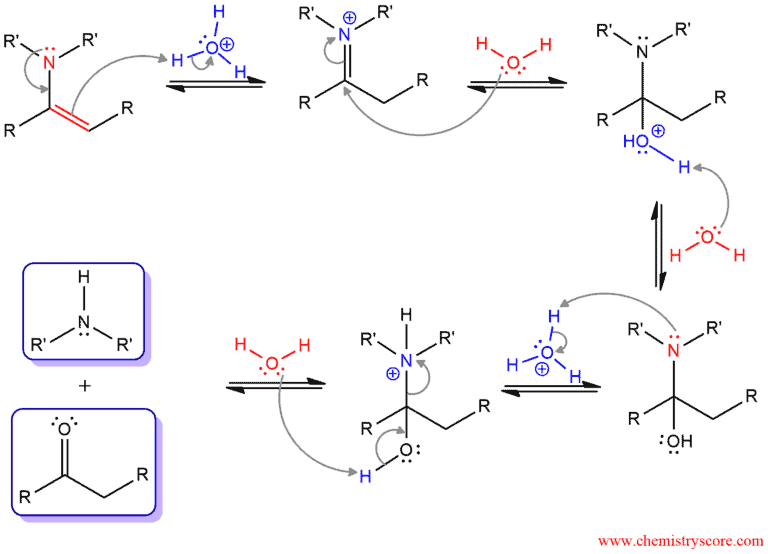
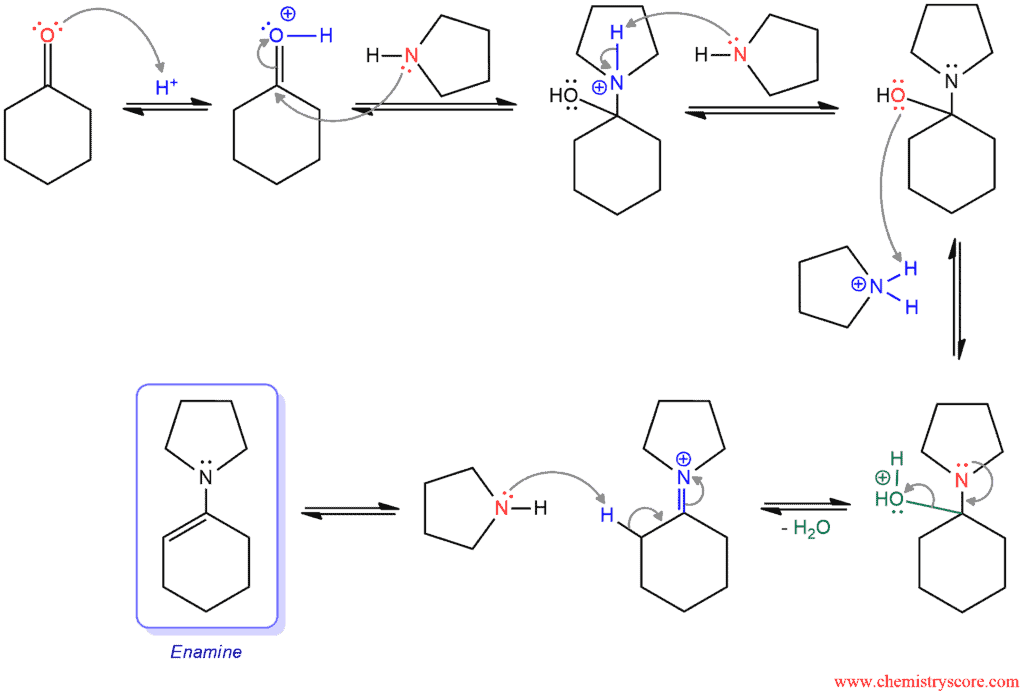
2/mechanism.png)