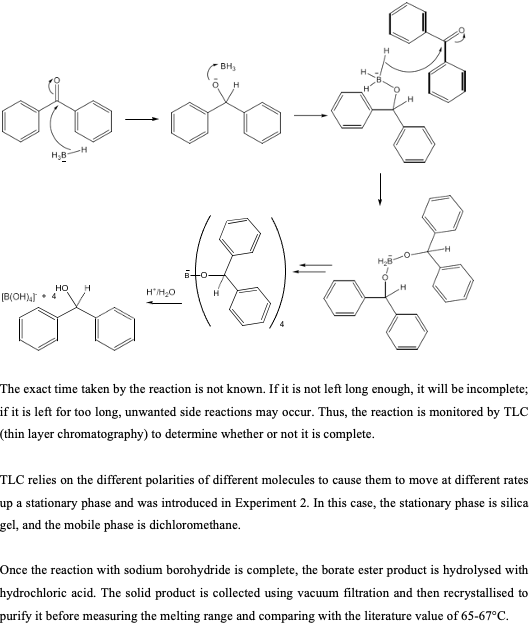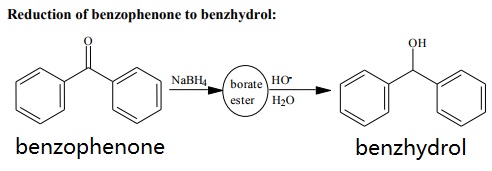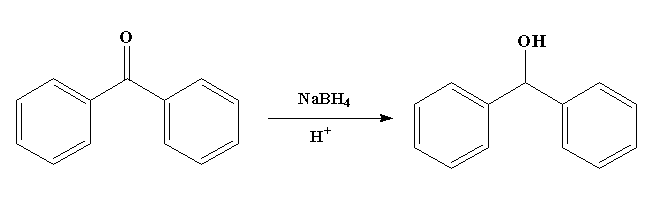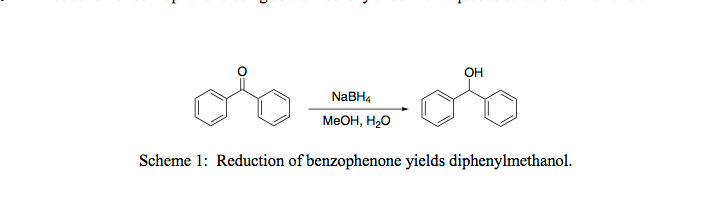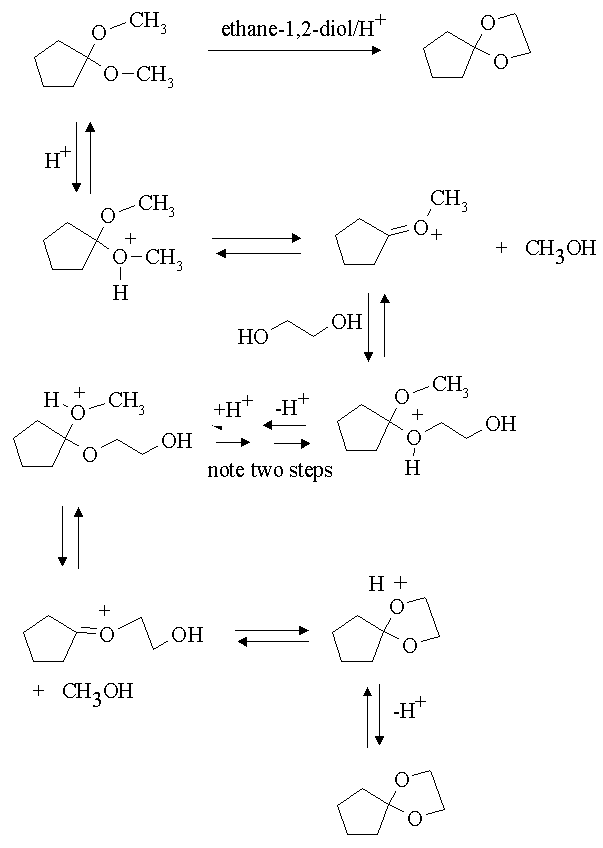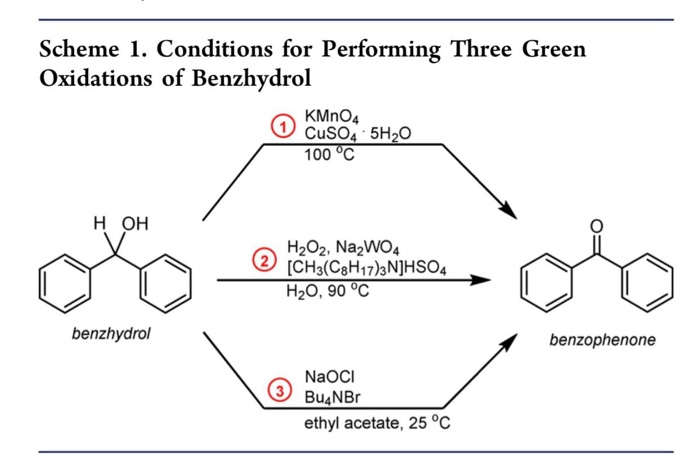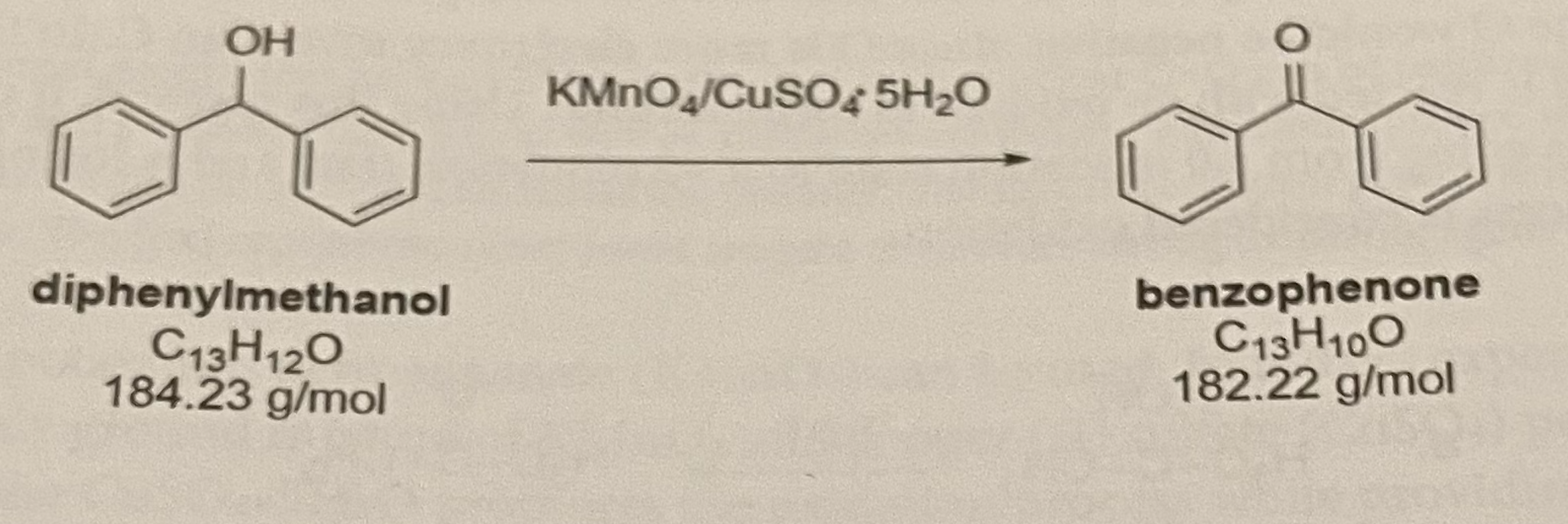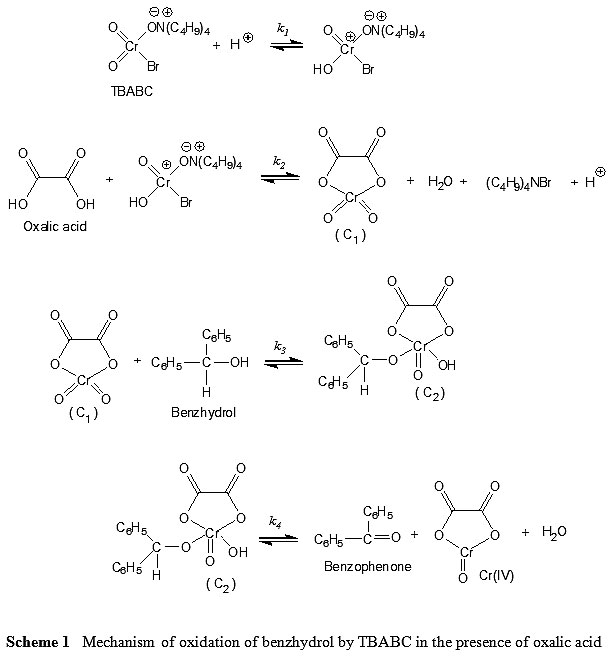
Kinetics and Thermodynamics of Oxidation of Benzhydrol by Tetrabutyl Ammonium Bromochromatein the Presence of Oxalic Acid : Oriental Journal of Chemistry

Oxidation of benzhydrol by tributylammonium chlorochromate: a kinetic and mechanistic study | SpringerLink
Molecules | Free Full-Text | A Straightforward Diphenylmethyl Protection Method and Deprotection of Some Pyrimidine Nucleosides | HTML

CHEM 2020 Textbook Notes - Fall 2017, Chapter 1 - Phenylmagnesium Bromide, Alcohol, Organic Synthesis

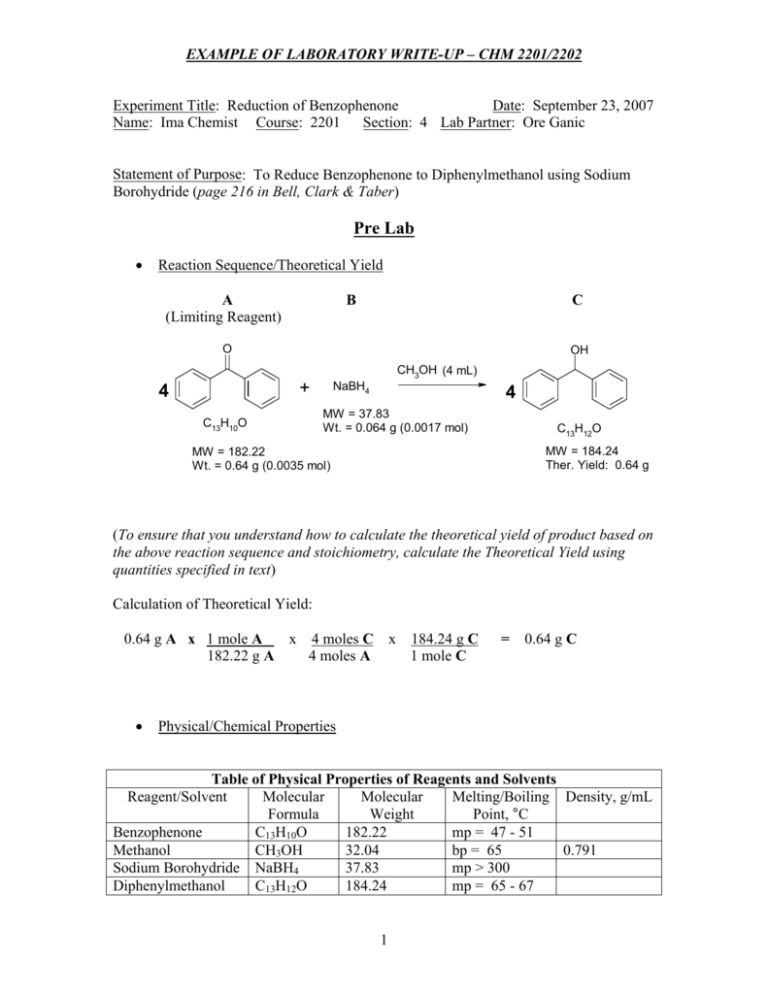
SOLVED:Page 102 Exp. 18: Reduction of Benzophenone After thic reaction neriod add \( 2 \mathrm{~mL} \) of water and heat intermittently with swirling in a hot water hath for 5 minutes Do

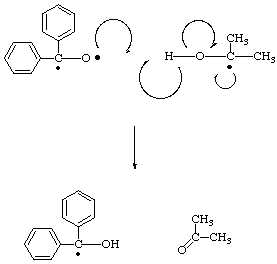
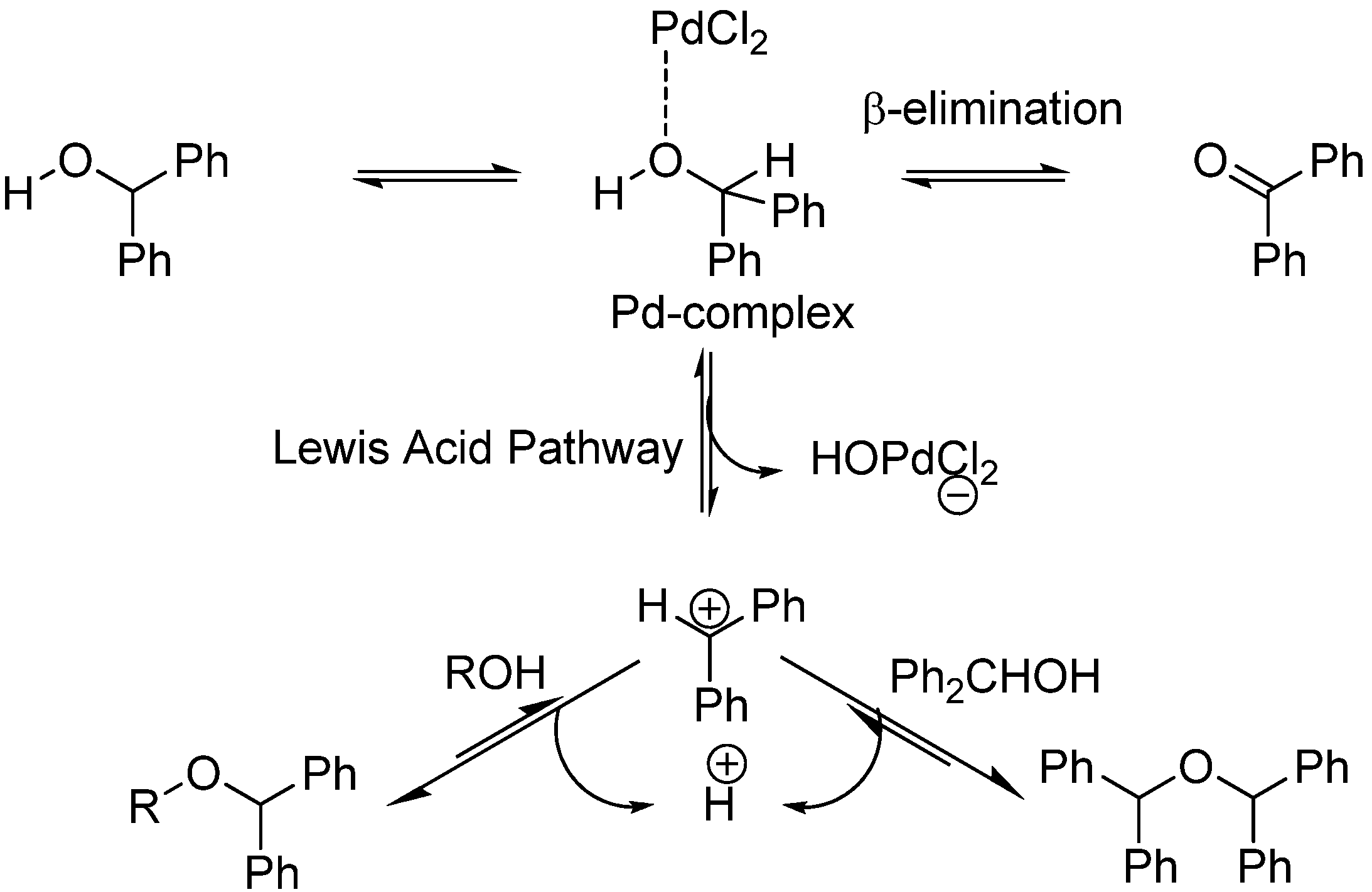
Exploring the mechanism of diphenylmethanol oxidation: A combined experimental and theoretical approach - ScienceDirect
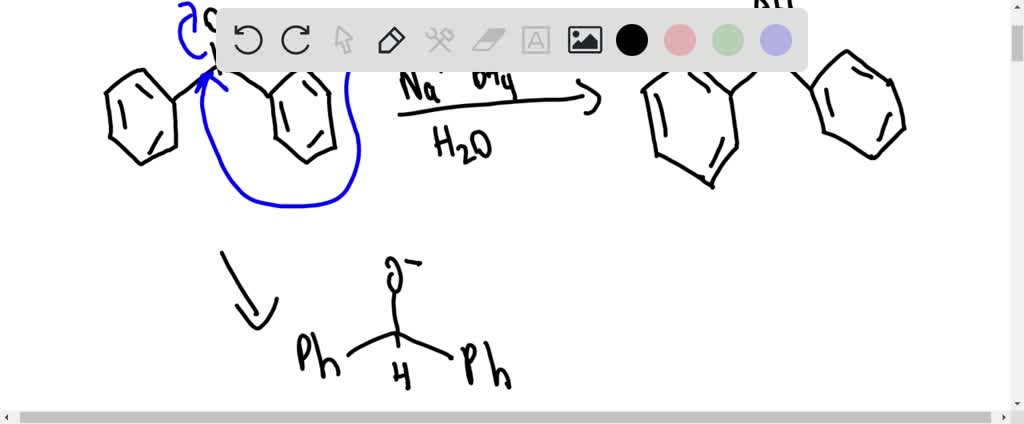
SOLVED:Page 102 Exp. 18: Reduction of Benzophenone After thic reaction neriod add \( 2 \mathrm{~mL} \) of water and heat intermittently with swirling in a hot water hath for 5 minutes Do

OneClass: Benzilic Acid Rearrangement lab: 1.) A side product formed in the benzilic acid rearrangeme...
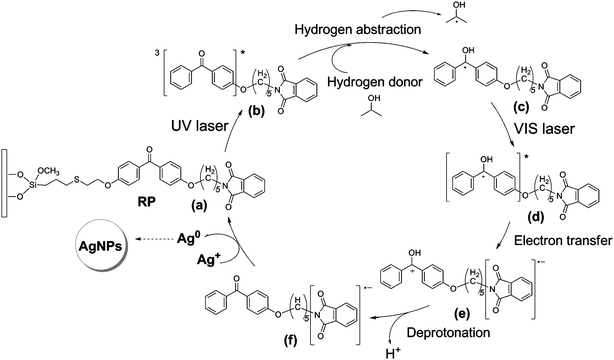
Photochemical fabrication of silver nanostructures at the solid–liquid interface using a recyclable photosensitized reduction process - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/B917981J
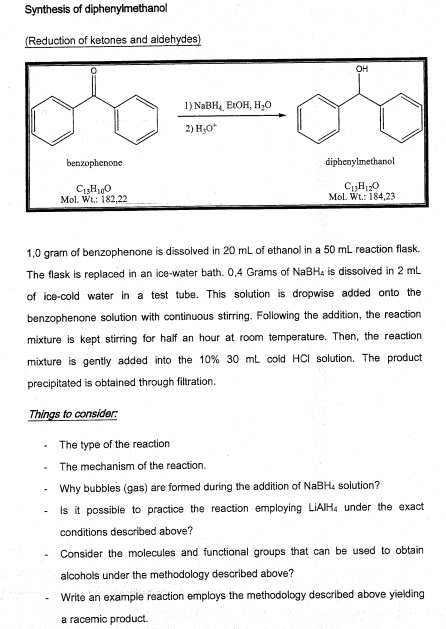
SOLVED:Synthesis of diphenylmethanol Reducten ketones and Idehxdes) 1) NaBl EtOH, H,o 2) Hjot hentubnanona dipbcovimathanol CuHjo MoL CwE,84,23 Mol Wt; 182,22 1,0 gram benzophenone d ssolved 20 ML ethano 50 mL reaction